IIAR White Paper: Environmental and Health Risks of Synthetic PFAS Refrigerants
TABLE OF CONTENTS
Executive Abstract
Executive Summary
Section 1: The Risk of HFO Refrigerants
Section 2: TFA: The Most Abundant PFAS
Section 3: TFA Health Effects: Reproductive Toxicity
Section 4: Health Impacts of PFAS Chemicals
Section 5: PFAS and TFA Pose a Planetary Threat to Water, Critical Ecosystems
Section 6: TFA Sources Must be Limited Now to Avoid Irreversible Contamination
Section 7: U.S. Regulations on PFAS Chemicals and TFA
Section 8: European Regulations on PFAS Chemicals and TFA
Section 9: Removal and Remediation Methods for TFA Removal from Water
Appendix 1: PFAS and TFA: Regulatory Landscape and Strategic Refrigerant Choices
References Appendix
Authors:
John Flynn
Tim Cook
Stephanie Smith
Andrea Fischer
EXECUTIVE ABSTRACT
This white paper examines the growing global concern over per- and polyfluoroalkyl substances (PFAS), focusing on trifluoroacetic acid (TFA), a persistent byproduct of modern hydrofluoroolefin (HFO) refrigerants. While designed to reduce global warming potential (GWP), HFOs degrade in the atmosphere to form TFA – a short-chain PFAS that accumulates in water and soil. TFA’s resistance to degradation and lack of large-scale remediation methods make it a mounting ecological and regulatory challenge.
This report outlines the sources, pathways, and impacts of TFA contamination, the limitations of current U.S. and EU regulatory frameworks, and the strategic imperative for industries to transition toward natural refrigerants such as CO₂, ammonia, and hydrocarbons. The analysis concludes that while HFOs achieve climate goals, they introduce long-term PFAS pollution risks requiring urgent source controls, regulation, and investment in non-PFAS alternatives.
EXECUTIVE SUMMARY
Hydrofluoroolefin (HFO) refrigerants, developed to meet low-GWP targets, have unintentionally become major contributors to PFAS contamination. As they degrade, HFOs transform into trifluoroacetic acid (TFA) – a highly mobile, persistent, and unregulated PFAS compound now detected globally in water, soil, food, and even human blood. TFA’s ubiquity poses uncertain risks to health, ecosystems, and long-term water security. The HFO refrigerants were engineered for lower GWP.
As HFC refrigerants are phased out, HFOs risk introducing new PFAS pollution. European data show HFO-1234yf could raise TFA in rainwater 250-fold if it replaces HFC-134a; U.S. sampling in 2023 detected TFA in 95% of Indiana drinking water samples. In Germany, TFA accounts for over 90% of total PFAS mass in drinking water and has increased fivefold since 2005.
TFA is currently classed as “low to moderate” in acute toxicity, but new studies link it to reproductive harm, prompting the European Chemicals Agency (ECHA) to propose its classification as toxic for reproduction, category 1B. Despite widespread detection, no defined U.S. health thresholds or regulations exist. New studies have shown that PFAS is bioaccumulative across generations, i.e. animals and potentially human offspring are born with their parents’ full PFAS exposure levels. This means that levels of PFAS (including TFA) being measured today may be much lower than actual exposure.
Despite increasing contamination levels, there are no currently established health thresholds defined to evaluate the threat of increasing TFA contamination in the U.S., or efforts to regulate their sources. TFA is the most abundant PFAS detected in water, soil, food and even the human body, and the HFO refrigerants that produce TFA are one of the leading sources of unregulated TFA contamination. As HFC refrigerants are phased out around the world, HFO refrigerants pose a major new PFAS contamination risk if they are adopted to replace HFCs.
Key Takeaways for Industry Stakeholders:
Regulatory Pressure: PFAS—including short-chain derivatives like TFA—are under growing global scrutiny. The HFO refrigerant market’s rapid expansion (~$3.6 billion by 2032) implies accelerating contamination risk.
Environmental Persistence: PFAS and TFA are highly stable, can be bioaccumulative, and are resistant to degradation. TFA accumulates in water systems and is extremely difficult to remediate. TFA is not produced naturally, but rather is the terminal breakdown product of larger PFAS chemicals, including HFO refrigerants.
Health and Ecological Risk: PFAS exposure is associated with cancers, reproductive toxicity, and potential planetary boundary threats.
Corporate Risk: Ongoing use of PFAS-based refrigerants increases legal and reputational exposure.
Natural Refrigerants: CO₂, ammonia, and hydrocarbons offer zero-PFAS solutions with minimal GWP and align with
sustainability mandates.
Future-Proofing: Transitioning to natural refrigerants now reduces energy use, regulatory burden, and long-term liability.
1. THE RISK OF HFO REFRIGERANTS
Introduction:
To meet low Global Warming Potential (GWP) targets, industry has developed hydrofluoroolefin (HFO) refrigerants and HFO/HFC blends. These compounds achieve lower GWP by combining a low-GWP HFO component with an HFC fraction that moderates flammability. However, all contain or generate per- and polyfluoroalkyl substances (PFAS) either directly or through atmospheric transformation.
What is PFAS:
PFAS comprise a broad category of chemicals including thousands of synthetic chemicals, many of which have been used since the 1940s in coatings, textiles, packaging, and firefighting foams. Their carbon–fluorine bond—the strongest in organic chemistry—makes them extraordinarily stable and resistant to degradation, earning the name “forever chemicals.”
These compounds are now ubiquitous, entering air, water, and soil through manufacturing, consumer use, and disposal. In refrigeration systems, industry standard annual leak rates of 10–20% release PFAS-bearing gases directly into the atmosphere. According to commercial refrigeration group, NASRAC, [supermarkets] leak on average 25% of the refrigerant charge each year. Meanwhile, the U.S. EPA sets “trigger rates” for leak repair requirements in commercial refrigeration at 20% and industrial process refrigeration at 30% per year. Comfort cooling systems have a 10% trigger rate. The newer HFC/HFO blends designed for low GWP standards, therefore, increase PFAS emissions through both leakage and intentional venting.
With the emergence of new HFC/HFO blends of refrigerants to meet GWP requirements, even more PFAS chemicals will be introduced into the environment directly through refrigerant leaks and intentional releases. In addition, these new chemicals may have unknown potential impacts, as it will take some time to thoroughly study their safety for common use.
HFO Refrigerant’s Lower GWP Results in Higher PFAS Release:
While HFOs reduce direct GWP, they convert almost completely into TFA within days—producing 5–33 times more TFA than legacy HFC-134a. European modeling shows that replacing HFC-134a with HFO-1234yf could raise TFA in rainwater by 250-fold.
This trade-off replaces climate impact with the enduring presence of PFAS pollution. Although HFOs serve as transitional options for existing R-134a systems, their atmospheric breakdown results in the long-term accumulation of TFA in water systems.
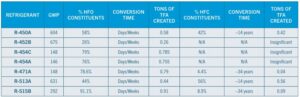
The following table was created to demonstrate the magnitude and speed at which refrigerant releases would occur in TFA contamination of our environment and waters for the newer HFO/HFC blends that have been developed to comply with lower GWP regulations.
The chart below provides a more comprehensive view of the GWP versus the conversion rate into TFA within the first two weeks of release into the atmosphere.
Conclusion:
HFO refrigerants meet short-term climate goals but generate TFA, a persistent PFAS resistant to water treatment. Complete atmospheric conversion and long environmental half-life make TFA a cumulative contaminant. Comparative data show HFOs yield far more TFA than HFCs, raising serious ecological and regulatory concerns. The challenge ahead is balancing climate benefits with chemical persistence to avoid replacing one global pollutant with another.
2. TFA: THE MOST ABUNDANT PFAS
Introduction:
TFA’s extreme persistence stems from its resistance to chemical, biological, and photolytic degradation. Once formed, it remains indefinitely, spreading rapidly through the global water cycle due to its high solubility and mobility. Rainwater in Germany, for example, now averages 335 ng/L TFA—a five-fold increase since 2005—and over 79% of European water samples contain measurable levels. It also escapes standard wastewater and drinking-water filtration, accumulating in rivers, groundwater, and ultimately oceans.
The pervasive contamination of water supplies and TFA’s bioaccumulative nature lead to an irreversible accumulation effect. Model projections suggest that TFA levels will continue to rise due to ongoing emissions from refrigerants, pesticides, and industrial sources. As it infiltrates groundwater, rivers, and drinking water supplies, TFA is absorbed into plant and animal food sources, as well as directly into humans through the water. Consequently, studies have found detectable levels of TFA in 98% of human blood samples. In addition to water supply contamination, TFA accumulates in soil, crops, and wild plants, resulting in soil concentrations exceeding 0.83 mg/kg (NOEC for terrestrial plants), which risks disrupting plant growth and the soil microbiome. TFA has been linked to reproductive toxicities and liver damage.
TFA has low acute toxicity for aquatic ecosystems but imparts chronic effects on algae and may potentially have similar effects on invertebrate species, although this is understudied.
What is TFA and why is it important:
TFA (CF₃COOH) is a synthetic, colorless, corrosive liquid with strong acidity derived from full fluorination of acetic acid. It is an ultrashort chain perfluoroalkyl acid (PFAA) within the PFAS family. Its high polarity and small molecular size make it exceptionally mobile in water and difficult to capture or degrade—hence its classification as a “forever chemical.” TFA’s physical properties are that it is a volatile liquid, which is highly soluble in water and other organic solvents.
HFO Refrigerants will Accelerate TFA Contamination:
HFOs react in the atmosphere—primarily with hydroxyl (OH) radicals—to form trifluoroacetyl fluoride, which hydrolyzes within 10–14 days to TFA. Studies show a nearly 100% conversion of released HFOs to TFA under normal conditions (Kazil et al., 2022).
Once formed, TFA dissolves in rainwater and accumulates in surface and groundwater systems. It eventually becomes absorbed through rain or direct contact into surface water and ultimately into drinking water aquifers. It will persist in the water system and bioaccumulate in organisms drinking from that water system. Due to the high polarity of TFA, it accumulates in water bodies, and with its resistance to degradation, the half-life of TFA in water is up to 30 years.
Furthermore, conventional water treatment methods are unable to remove TFA. New methods are being considered, but are costly and technically difficult at scale, which will be addressed in a separate section of this white paper.
Conclusion:
TFA’s dominance among PFAS arises from its multiple industrial precursors, environmental stability, and absence of natural degradation. Beyond refrigerants, PFAS-containing pesticides, pharmaceuticals, and fluoropolymer manufacturing also emit TFA precursors.
Because no large-scale removal methods exist, TFA concentrations are projected to rise globally. Reverse osmosis can capture it, but this method is too costly for municipal use. Without source control, TFA will continue to accumulate, posing a threat to long-term water and food security.
3. TFA HEALTH EFFECTS: REPRODUCTIVE TOXICITY
Introduction:
Although TFA exhibits relatively low acute toxicity, increasing concentrations and emerging evidence of developmental toxicity in animals have raised alarm. Broader PFAS research connects exposure to hormonal disruption, infertility, and developmental delays. Scientists warn that PFAS, including TFA, contamination may contribute to exceeding planetary boundaries for safe chemical exposure—thresholds beyond which human and ecological systems could be irreversibly harmed.
The pervasive presence and rising concentration levels have led scientists to propose that TFA pollution may represent a coming breach of planetary boundaries for safe chemical exposure. Planetary boundaries refer to the levels of synthetic chemicals, plastics, and other human made substances that, once reached, can disrupt ecosystems and human health, including human and animal fertility.
What is known about PFAS exposure points to hormonal and reproductive effects. For instance, PFAS can interfere with natural hormones, leading to thyroid dysfunction and reproductive issues, which include decreased fertility, pregnancy induced hypertension, preeclampsia, and developmental delays in children. Furthermore, changes in puberty timing, bone variations, and behavioral changes have been observed in children with elevated PFAS exposure. Taken together, these observed effects on fertility and developmental impacts of PFAS chemicals – that are already being observed at contamination levels much lower than projected over the coming decades – make PFAS exposure a particular existential risk to human health.
The bioaccumulative effects of PFAS are now known. A pivotal study by Pickard et al. (2022), titled “Maternal Transfer and Tissue Distribution of Per- and Polyfluoroalkyl Substances (PFAS) in Mice Following Exposure to Contaminated Water,” investigated how PFAS compounds are transferred from mother to offspring and how they accumulate in tissues across generations. For example, the pups of mice exposed to PFAS will inherit both their mother’s baseline PFAS levels and any accumulation they incur during their lifetime.
The Pickard study highlights the chilling fact that the levels of human exposure to PFAS – that are being studied now – may not be accurate indicators of PFAS exposure in subsequent generations. Due to maternal transfer of PFAS, generations being born today will not only start with higher PFAS exposure than their parents but may also accumulate significantly higher PFAS levels in their lifetimes than those being measured today.
The challenges faced now with PFAS can be compared to the discovery of endocrine disrupters a decade ago. Scientists were then focused on chemicals like bisphenol A (BPA), phthalates, polychlorinated biphenyls (PCBs), and nonylphenols—compounds found in plastics, pesticides, and industrial byproducts—that could interfere with hormonal systems. These endocrine-disrupting chemicals (EDCs) have been shown to mimic or block natural hormones, resulting in developmental, reproductive, neurological, and immune effects in both wildlife and humans.
Studies highlighted particular concern for vulnerable populations, especially children, due to their heightened sensitivity during critical growth periods. The early 2000s marked a turning point in recognizing EDCs as a global public health issue, prompting calls for regulatory scrutiny and further investigation into long-term impacts.
Establishing Exposure Thresholds:
Thousands of PFAS compounds remain unregulated and untested for blood concentrations or toxicity. The US EPA asserts that no safe level of PFAS in blood has been established.
The European Chemicals Agency (ECHA) proposes classifying TFA as toxic for reproduction (Category 1B) and very persistent and very mobile (vPvM). This process could take years, but it reflects a growing consensus that TFA’s toxicity is underestimated.
For now, safe TFA exposure levels must be extrapolated via comparison with data available for PFAS generally, a concerning task given that there is no absolute safe level of PFAS concentration in the blood, according to the US EPA. The EPA notes that adverse health effects may occur at any detectable concentration level, similar to other toxicants, such as lead or benzene.
Although this classification process is expected to take several years, it anticipates the potential that TFA may eventually be determined to be much more toxic than its current classification, which may be low simply because TFA contamination levels and resulting health effects have not yet reached the projected levels.
Barriers to Reducing Exposure:
Reducing exposure is complicated by industrial resistance and regulatory inertia. The Make America Healthy Again Report (2024) revealed that chemical companies spent over $77 million lobbying in 2024, much of it aimed at suppressing PFAS research and delaying regulation echoing historical tactics employed by the tobacco industry.
Nevertheless, these synthetic chemicals pose a potentially serious threat to human health, and the industries that produce them represent an additional barrier to raising awareness of their risks, thereby reducing the time to ban their sources before they can irreversibly accumulate in the human body and environment.
General PFAS Exposure Measured in Human Blood:
Nearly all people worldwide have measurable PFAS in their blood. CDC data show >99% of Americans carry at least one PFAS compound. Median serum levels (NHANES) are 4.3 ng/mL (PFOS) and 1.47 ng/mL (PFOA). Highly exposed workers once reached >1,000 ng/mL.
While TFA-specific data are limited, available evidence suggests TFA is less bioaccumulative than long-chain PFAS, though it remains highly persistent and mobile in the environment. Although TFA is not known to bioaccumulate, its water solubility and prolific presence in water, plants and animals, will introduce new TFA to replace any that the human body may shed, thus having a similar effect to bioaccumulation.
The National Academies of Science (2022) recommends 2 ng/mL as a clinical action level for PFAS—not a safety threshold. According to the Montreal Protocol’s Environmental Effects Assessment Panel (EEAP), TFA poses a negligible risk to human health and the environment, but according to the panel, many scientists disagree, arguing that even in the absence of evidence of toxicity, persistence alone is a significant cause for concern.
The EEAP acknowledged that its recent 2024 update does not reflect the ongoing scientific debate around TFA, particularly with respect to its persistence, and despite the concerns about TFA raised by many in the scientific community, the fluorochemical industry continues to promote the narrative that TFA is a safe, naturally occurring substance and thus HFOs pose no risk in this regard. According to EEAP, the industry often omits any reference to unfavorable evidence or scientific interpretations that would bring this into question, and many papers on this topic have been written, co-written or funded by the fluorochemical industry.
While most people in the US have PFAS levels below 5 ng/mL, there are two categories of citizens with significantly higher concentration levels in their blood serum. First, people with occupational exposure, such as firefighters or workers in the PFAS manufacturing process, have much higher levels. For example, in 2000, highly exposed workers in the manufacturing process had average PFOA blood levels exceeding 1,000 nanograms per milliliter (ng/mL).
The second category of elevated exposure is for communities with contaminated water supplies, where the median PFAS concentration levels are 2-3 times higher than those of the rest of the population. It has been demonstrated that due to the persistence of PFAS exposure and stability, PFAS concentration levels remain in the human body for years. That said, it has also been shown that levels will decline over time if exposure is reduced. Currently, blood testing provides insight into exposure history and subsequent blood serum concentrations, but is not yet effective for diagnosing specific health outcomes.
Despite this fact, the 2022 National Academies of Sciences, Engineering, and Medicine (NASEM) report provided practical clinical guidance stating that adverse health effects related to PFAS exposure are not expected at concentration levels below 2 ng/mL in blood serum. It is important to note that this 2 ng/mL threshold is a clinical action level intended to guide medical counseling and monitoring – it is not a regulatory limit or enforceable standard.
However, above this level, the potential for health effects increases, and individuals may benefit from exposure reduction counseling. Notably, the threshold level is significantly below the population median level, indicating that most Americans, even those not in higher-risk occupations or communities with water contamination, may benefit from exposure reduction. For additional health-based guidance values, the European Food Safety Authority (EFSA) and the HBM4EU project use a value of 6.9 ng/mL as a reference for increased risk in the general population.
Conclusion:
PFAS, including TFA, are nearly universal in human blood. Some legacy compounds are declining, but unregulated PFAS, such as TFA, are rising. Given its persistence and emerging reproductive risk, TFA warrants urgent study and regulation. While concentrations have declined for some regulated compounds, such as PFOS and PFOA, due to regulatory actions and voluntary phase-outs, many other PFAS, including TFA, remain unregulated and understudied. Blood serum levels vary significantly depending on occupational exposure and environmental contamination. While no level is considered entirely safe, clinical guidance suggests that health risks increase above 2 ng/mL. The persistence of PFAS in the human body and the environment underscores the importance of continued monitoring, research, and regulatory efforts. As new PFAS compounds are introduced and detected, understanding their health implications and exposure pathways will continue to be a critical public health priority.
4. HEALTH IMPACTS OF PFAS CHEMICALS
Introduction:
Extensive research links PFAS exposure to cancer, immune suppression, metabolic disorders, and developmental effects. And studies have confirmed that PFAS disrupt hormones, metabolism, and immune regulation. Moderate associations have been observed with thyroid dysfunction, changes in liver enzymes, reproductive and developmental outcomes, and kidney disease. Additional research suggests possible connections to metabolic disorders, asthma, and neurobehavioral changes, although findings in these areas are less consistent. Studies support these associations through various pathways, including endocrine disruption, immune modulation, oxidative stress, and epigenetic alterations.
Exposure Effects:
PFAS chemical exposure has been linked to increased risk of contracting certain kinds of cancer, including kidney, testicular, and prostate cancers. In fact, the specific PFAS and common byproducts of other PFAS chemicals, Perfluorooctanoic Acid (PFOA) and Perfluorooctane Sulfonate (PFOS), have been classified as carcinogenic to humans by the World Health Organization, based on molecular effects and epidemiological evidence.
PFAS chemical exposure is also linked to reduced immune function. This includes lower antibody responses to vaccines, which consequently lead to an increased incidence of infection, especially in children. Furthermore, there is evidence that PFAS exposure can increase the risk of atopic dermatitis and of lower respiratory tract infections for children exposed prenatally or during infancy.
PFAS has been found to contribute to metabolic and liver effects. PFAS exposure has been consistently linked to elevated cholesterol levels and alterations in liver enzymes. There is further evidence linking PFAS to metabolic syndrome, obesity, and both Type 1 and gestational diabetes.
Studies have also shown that PFAS exposure contributes to diminished kidney function, increased serum uric acid concentrations, which can lead to gout and kidney stones, and a higher risk of chronic kidney disease, both in adults and children.
PFAS exposure has been shown to lead to developmental effects during pregnancy, including decreased birth weight and developmental delays in children.
Additionally, associations with asthma and neurobehavioral changes have been reported; however, the findings are inconsistent and may depend on factors such as age, sex, and others. Similarly, some studies suggest links to cardiovascular disease and related risk factors.
The following table summarizes the health impact linkages and strength of the association:
Several mechanisms have been found to explain the association and contributing factors that PFAS exposure may have
with cancer development. They include the following:
Endocrine Disruption: PFAS are recognized as endocrine-disrupting chemicals. They can interfere with hormone regulation, which is particularly relevant for hormone-sensitive cancers such as thyroid, breast, and prostate cancer.
Epigenetic Changes: PFAS exposure can alter DNA methylation and histone modification patterns, potentially activating oncogenes or silencing tumor suppressor genes—key processes in the initiation and progression of cancer.
Chronic Inflammation and Immune Suppression: PFAS can induce chronic inflammation and suppress immune function, both of which are known to contribute to cancer risk by impairing the body’s ability to detect and destroy abnormal cells.
Oxidative Stress and Disruption of Cell Metabolism: PFAS may cause oxidative stress and disrupt normal cellular metabolism, further increasing the risk of mutations and uncontrolled cell growth.
Binding to Nuclear Receptors: PFAS can bind to nuclear receptors such as peroxisome proliferator-activated receptors (PPARs), which regulate lipid metabolism and are implicated in cancer development.
Finally, research has suggested that PFAS exposure may double the likelihood of prior cancer diagnoses in women, particularly melanoma and uterine cancer. Some PFAS compounds have demonstrated stronger associations for cancer risk in females, possibly explained by hormonal differences. In addition to stronger linkages to females, children also may be more susceptible to PFAS-related health effects due to relative exposure rate per body weight and developmental vulnerability.
Conclusion:
Given the persistence of PFAS in the environment and their widespread detection in human populations, regulatory scrutiny has increased. While existing regulations address some PFAS compounds, the breadth of emerging health data suggests that more comprehensive and stringent regulations are likely to be implemented in the future.
5. PFAS AND TFA POSE A PLANETARY THREAT TO WATER, CRITICAL ECOSYSTEMS
Introduction:
HFO refrigerants, designed for low GWP, are a significant source of PFAS. Their atmospheric degradation produces trifluoroacetic acid (TFA), which accumulates globally in water.
Without rapid intervention, TFA concentrations in water are projected to rise by 10-to 100-fold by 2040, exceeding all existing safety thresholds. A combination of bans on primary TFA sources, such as HFO refrigerants, binding limits, and innovation funding, offers the best path to curb this irreversible contamination.
Water Supply Impacts:
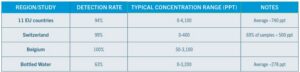
The rapid HFO degradation leads to regional TFA accumulation, whereas the more protracted HFC-derived TFA disperses globally, though both contribute to TFA contamination. In Europe, 94% of tap water and 63% of bottled water contain detectable TFA, with some levels up to 4,000 ppt.
Because the HFO refrigerant components degrade readily within 2 weeks of release into the atmosphere, HFO releases have a regional impact on TFA, whereas HFC-derived TFA disperses globally due to its longer atmospheric lifetime, contaminating remote regions, including the Arctic. The TFA in the atmosphere can be deposited through rain or snow via a wet deposition mechanism or through direct contact onto land or water surfaces via a dry deposition mechanism.
The aquatic accumulation of deposited TFA occurs through wet and dry deposition onto surface water, or indirectly into groundwater through dry deposition onto surface soil. The flow of surface water into the oceans makes the ocean the terminal sink for TFA, such that deep—water concentrations can persist for centuries.
Concentration levels in the EU study ranged from undetectable to 4,000 parts per trillion (ppt), with averages between 740 ppt and 770 ppt. As a reference point, the EPA’s national drinking water standards set a maximum contaminant level (MCL) for longer-chain PFAS (PFOA and PFOS) at 4 ppt. The following table offers a broader review of the data from the EU tap water study:
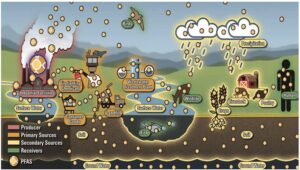
Sources of Water Contamination:
In addition to HFO refrigerants and some HFC refrigerants, many PFAS-containing pesticides also degrade when released
into the environment, resulting in the formation of TFA, making agriculture a significant source of TFA in rural water
supplies. These pesticides, such as flufenacet, are applied to millions of hectares of farmland worldwide, providing a
continuous input of TFA into our water supplies. Similarly, HFO and some HFC refrigerants used in refrigeration systems
worldwide provide a nearly constant source of fugitive emissions, which degrade into more TFA, ultimately being
absorbed into the water supply. Finally, there are also direct emissions from industrial manufacturing processes, such
as pharmaceuticals and fluoropolymer production, as well as the release of PFAS chemicals or TFA precursors during
production.
Source: USGS
Eco System Contamination:
TFA’s persistence and solubility enable global spread, affecting rainwater, crops, and oceans. It inhibits algal growth, disrupts aquatic food webs, and alters soil nutrient cycles, thereby reducing agricultural productivity. Because TFA has been detected universally in ground and surface waters, and it is resistant to current water treatment filtration, TFA infiltrates all water supplies before terminally reaching and contaminating ocean waters.
TFA does not bioaccumulate in humans or animals, unlike in plants, although it is detectable in approximately 98% of the blood samples taken in the U.S.
TFA has been found to accumulate in crops, tea, and beer, creating an additional indirect exposure pathway to humans through the food chain. Additionally, the prevalence of TFA in surface water has been found to impact aquatic ecosystems. Most concerning, it has been classified as harmful to marine life because TFA disrupts microbial and plant growth in water systems, the health of which underpins critical food-producing ecosystems.
For aquatic ecosystems, TFA inhibits growth in algae at low concentrations, disrupting the base food-web dynamics of the ecosystem. Marine diatoms exhibit reduced photosynthetic efficiency, which may also impact carbon sequestration, thereby exacerbating climate concerns.
As with humans, most aquatic organisms exhibit no acute toxicity from TFA at current levels, but may show chronic impacts on reproduction, behavior, or endocrine systems over time, although this is understudied. In the terrestrial ecosystem, TFA concentrations in the soil are increasing and demonstrably bioaccumulate in crops at higher rates than other PFAS chemicals. This threatens the food chain, creating exposure pathways for herbivores and humans.
No studies have yet been conducted to explore the long-term effects on pollinators or soil invertebrates. However, TFA does alter the nitrogen and phosphorus cycling in soils, potentially reducing the agricultural productivity of the soil. TFA presents a nearly impossible challenge due to its irreversible consequences for both aquatic and terrestrial ecosystems. Its extreme persistence and solubility in water ensure perpetual accumulation with no natural degradation pathways. Furthermore, remediation is currently stymied by a lack of effective large-scale water treatment methods.
Conclusion:
TFA meets all three planetary boundary threat criteria: persistence, mobility, and ecological disruption. Without immediate emission limits, irreversible contamination of global water systems will continue. TFA has disruptive effects, including algal inhibition and soil toxicity, which have already been observed. Because of the demonstrated global impacts due to TFA’s ubiquity in water supplies, rainwater, arctic ice, and plants and animals, additional study and understanding of TFA is critical. TFA contamination produces irreversible effects due to its resistance to degradation, and the lack of cost-effective remediation methods.
6. TFA SOURCES MUST BE LIMITED NOW TO AVOID IRREVERSIBLE CONTAMINATION
Introduction:
PFAS chemicals persist across air, soil, and water. Once released, they contaminate food chains. TFA’s solubility and resistance to filtration make accumulation inevitable unless sources are curtailed.
Once in the soil, PFAS can alter soil chemistry and be absorbed into microbial communities and plant material, introducing the contaminant into the food chain. Similarly, PFAS chemicals dissolve into water, making it very difficult to remove because traditional filtration methods are ineffective at removing PFAS. The chemicals contaminate and accumulate in rivers, lakes, and groundwater, which directly introduces the chemicals into our water supply and, by extension, into the food chain.
With multiple and wide-ranging contamination paths, and due to their chemical stability and resistance to degradation, PFAS chemicals are rapidly accumulating in the atmosphere, soil, water, and in humans and other animals within the food chain. Without swift action to limit the sources of this contamination, today’s TFA pollution will become tomorrow’s existential threat to human health and planetary survival.
The Need for TFA Source Bans:
The long-term implications of TFA accumulation are profound. Scientists warn that TFA’s irreversible accumulation may disrupt the Earth’s chemical equilibrium, posing a threat to the planetary boundary. Even if governments and countries were able to immediately and effectively halt emissions, legacy pollution, which currently exists in environmental TFA levels, will persist for millennia. With existing PFAS remediation approaches that often generate TFA as a byproduct, the TFA problem appears to be intractable.
Whether intractable or not, the consequences of inaction are too severe to consider. Immediate action is required to curtail the primary sources of TFA – PFAS pesticides and F-gases – which are under scrutiny, with calls for rapid bans or severe restrictions to prevent further contamination. An effective long-term solution will require source control, comprehensive monitoring, and updated regulatory frameworks that specifically address TFA, as well as broader PFAS pollution. Mitigation pathways under consideration include PFAS pesticide bans, F-Gas phaseouts, source-specific regulations targeting industrial TFA precursors, as well as experimental TFA destruction methods such as advanced oxidation.
Perhaps most importantly, regulating TFA concentration levels in tap water fails to address the sources of TFA. Without such source bans, TFA concentrations are expected to rise by 10-100 fold in just 15 years by 2040. The key challenges for the existing TFA problem, which will only be magnified over time, include removal, monitoring, and byproduct formation. TFAs’ high solubility and persistence, along with the ineffectiveness of current water treatment approaches, mean that there is no practical way to remove the chemical from drinking water.
Because TFA has historically been classified as a non-relevant metabolite, it has not been systematically monitored, leading to unobserved and widespread accumulation. Furthermore, many of the PFAS remediation technologies employed to address and remove longer-chain PFAS inadvertently generate TFA as a byproduct of breakdown, complicating cleanup efforts.
Governments Must Meet the Regulatory Challenge:
The OECD recognizes TFA as PFAS, but both the EU and the US lack consistent regulation. Denmark and the Netherlands have set guidance limits (900–2,200 ppt), but these remain voluntary. Furthermore, the US EPA’s 2024 PFAS drinking water standards specifically exclude TFA.
Still, comprehensive PFAS regulation is required under TSCA, REACH, and national water laws. The EU’s proposed REACH restriction seeks to phase out HFO and HFC refrigerants; In the U.S., Maine has already banned PFAS compounds, including HFOs.
TFA is not yet comprehensively regulated in drinking water in the EU, although ongoing scientific and political debates continue regarding its risk and the need for stricter controls. Because there are inadequate studies to determine the toxic effects on aquatic and terrestrial organisms, or the impact on soil biodiversity and nutrient cycles, regulators are reluctant to impose binding global limits for TFA in the US and elsewhere.
Governments remain slow to enact regulations, pending more definitive academic studies. The Organization for Economic Co-operation and Development (OECD), comprised of 38 countries from the Americas, Europe, Asia, and Australia, has classified TFA as a PFAS chemical. Even so, it is not currently regulated as strictly as other PFAS in the EU and US Drinking Water directives.
Standard water treatment methods do not alter TFA concentrations during water treatment. Current regulatory approaches are inconsistent and inadequate. The PFAS total limit of 500 ppt may not include or adequately address TFA, which often constitutes more than 98% of PFAS, and current levels in EU tap water studies already exceed this limit. Furthermore, the Danish and Dutch TFA-specific guidelines are inconsistent and fail to account for the cumulative, irreversible buildup of this “forever chemical.”
On a more positive note, PFOS concentrations in the US have declined by more than 85% since 2000, and PFOA concentrations have also declined, albeit by only 70%. These trends have been confirmed by multiple studies, which attribute the decline to regulatory actions and the phase out of PFOS and PFOA production and use since the early 2000s. Initially, growing international concern about the persistence and health risks of PFOS and PFOA led major manufacturers to voluntarily phase out production of these chemicals.
Complementing these voluntary actions, the US EPA used the Toxic Substances Control Act to restrict the manufacture, import, and use of certain PFAS. Then, in April 2024, the EPA finalized legally enforceable Maximum Contaminant Levels for PFOA and PFOS in drinking water at 4 parts per trillion, requiring public water systems to comply by 2029. The EPA has further utilized the Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA, also known as Superfund) to address and remediate PFAS-contaminated sites, while leveraging the Resource Conservation and Recovery Act (RCRA) and the Clean Water Act (CWA) for additional authorities to manage PFAS waste and water discharges.
Given the magnitude of the problem, significant government regulation will be required to contain the issue. If there are any lessons to be learned from ozone-depleting CFCs, it is that delaying action risks irreversible harm. In contrast, the success of definitive regulatory actions under the Montreal Protocol offers hope.
Governments will also need to overcome technical challenges to find cost-effective, large-scale remediation, while also addressing industry resistance from stakeholders involved in these businesses. Reducing TFA emissions is now essential to prevent its compounding damage to water security, agriculture, and ecosystems. Without intervention, TFA’s irreversible accumulation will burden future generations with this “forever chemical” crisis.
While slow to start, some governments are considering regulatory actions to stem the pervasive increases in TFA concentration levels in water reservoirs by regulating sources of PFAS and may require industry and municipalities to develop and deploy reduction or removal processes within their water treatment systems. In the EU, five EU countries (Germany, Denmark, Sweden, Norway, and the Netherlands) have proposed a REACH regulation ban on PFAS refrigerants targeting HFCs (R-134a, R-125, HFOs (R-1234yf, R-1234ze), and their blends.
In the US, federal actions on PFAS chemicals have been limited to PFOA and PFOS, specifically excluding TFA. However, several states, including Maine, are considering bans, with Maine having passed a prohibition on PFAS compounds.
Conclusion:
TFA’s persistence demands immediate source reduction. Past success in phasing out PFOS and PFOA demonstrates that decisive regulation can be effective. Acting now will protect water, agriculture, and ecosystems from irreversible contamination.
Although TFA does not bioaccumulate in humans, its presence in crops, beverages, and aquatic ecosystems raises concerns about indirect exposure and ecological impacts. Current regulatory frameworks vary significantly across regions and often do not explicitly address TFA, despite its prevalence and potential health risks. Projections indicating a 10- to 100-fold increase in environmental TFA levels by 2040 underscore the need for continued scientific assessment and the likely evolution of more immediate targeted and stringent regulatory responses.
7. U.S. REGULATIONS ON PFAS CHEMICALS AND TFA
Introduction:
US regulations target legacy PFAS, such as PFOA and PFOS, but thousands of related compounds—including TFA— remain unregulated. The OECD classifies TFA as a PFAS, but the US EPA does not.
Meanwhile, the definition of Trifluoroacetic Acid (TFA) as a PFAS chemical by the Organization of Economic Cooperation and Development (OECD) has not been adopted by the US Environmental Protection Agency (EPA), limiting the regulator’s appetite for regulatory actions addressing TFA releases and accumulation in US waters.
U.S. Regulations:
The US EPA leads federal efforts to regulate PFAS chemicals due to their environmental persistence and bioaccumulation. The federal agency exercises its authority under several congressional statutes, including the Toxic Substances Control Act (TSCA), the Safe Drinking Water Act (SDWA), the Clean Water Act (CWA), the Comprehensive Environmental Response, Compensation, and Liability Act (CERCLA), and the Resource Conservation and Recovery Act (RCRA).
In April 2024, the EPA finalized the first-ever drinking water standards for several PFAS under the CWA. The enforceable Maximum Contaminant Levels (MCLs) are set at 4 parts per trillion (ppt) for PFOA and PFOS individually, and 10 ppt for PFNA, PFHxS, and Gen X chemicals. Although the MCL was set at 4 ppt for PFOA and PFOS, the Maximum Contaminant Level Goal (MCLG) for these chemicals is zero, reflecting the latest science that asserts that no safe level of exposure to these PFAS chemicals exists. Furthermore, the EPA is developing new effluent discharge limits for PFAS under the CWA, targeting manufacturers and metal finishers to reduce the release of PFAS into waterways.
In April 2024, PFOA and PFOS were designated as hazardous substances under CERCLA (Superfund), enabling federal action for cleanup and liability for contamination. Then, as of 2025, the EPA requires manufacturers and importers to report detailed information on all PFAS manufactured or imported since 2011 under TSCA Section 9(a)(7), including production volumes, uses, worker exposure, and disposal practices.
The Toxics Release Inventory (TRI) was expanded to include more PFAS, now totaling 205 reportable compounds as of March 2025. Under the Significant New Use Rule (SNUR), manufacturers, must notify the EPA before resuming or starting new uses of 329 “inactive” PFAS, triggering a risk assessment before approval, which may delay or prevent the use of newer or inactive PFAS chemicals.
In addition to these federal activities, regulations have been enacted at the state level, which are often stricter than PFAS regulations. States like California, Minnesota, and Colorado are banning PFAS in various consumer products, including textiles, cookware, and cosmetics, starting in 2025. Minnesota is going further with an intended regulation to phase out all intentionally added PFAS in products by 2032, unless deemed unavoidable. Similarly, many states have set their own MCLs and cleanup standards for PFAS to bolster and/or strengthen federal drinking water standards.
As noted above, the US EPA excludes TFA from its working definition of PFAS due to structural criteria, environmental behavior, and regulatory prioritization. For the structural definition, TFA is excluded because the US EPA definition of PFAS requires at least two adjacent carbon atoms with at least one fully fluorinated and the other at least partially fluorinated. Because TFA is a single, fully fluorinated carbon molecule, it does not meet the strict EPA definition, although TFA is a terminal degradation byproduct of virtually all PFAS chemicals. The EPA excludes TFA for environmental reasons, citing that TFA is naturally occurring in oceans as a byproduct of reactions from deep-sea volcanic activity. However, this position is controversial and not universally accepted.
The EPA Outlook on TFA Regulation:
TFA has one fully fluorinated carbon, excluding it from the EPA’s structural definition. The EPA also cites minor natural formation and low bioaccumulation. However, most scientists confirm TFA is anthropogenic, primarily from HFCs, HFOs and pesticides.
For example, a 2023 study found that TFA was detected in 95% of the drinking water samples from Indiana. However, no established health thresholds have been defined to evaluate the threat posed by TFA levels.
With EPA’s narrower working definition of PFAS compared to international bodies such as the OECD and the EU’s REACH regulation, TFA’s exclusion means that it has no reporting requirements under the TSCA, no cleanup mandates under CERCLA, and no drinking water limits under the CWA. The following table summarizes the implications of TFA’s exclusion from the PFAS definition by the US EPA:
This definitional divergence has significant regulatory implications, particularly for substances like TFA that fall outside the EPA’s scope but are included under international frameworks.
The EPA Should Prioritize Regulation:
EPA may revise definitions as evidence grows. Advocates warn that exclusion would repeat the PFAS regulatory failures of the past. TFA remains outside the scope of current US PFAS regulations due to its chemical characteristics and risk profile. However, it is monitored as a byproduct of certain refrigerants and industrial processes. The regulatory landscape for PFAS is evolving rapidly, with ongoing federal and state actions aimed at reducing human and environmental exposure.
The industry has made strong statements opposing TFA’s classification due to structural differences in TFA, as well as its natural occurrence. Industry groups cite studies that show minimal confirmed health risks and a lack of bioaccumulation. However, TFA is highly mobile in water.
These industry groups have clear economic motivations, as classifying TFA as a PFAS would threaten lucrative markets for refrigerants and pesticides, and expose companies to regulatory costs for water treatment upgrades, cleanup, and reporting. These arguments are drawing criticism from environmental lawyers like Rober Bilott, who warns that dismissing TFA risks repeating the PFAS crisis, where delayed action led to widespread contamination. Emerging studies showing the ubiquity of TFA in water supplies and inconsistencies in definitions with international bodies also threaten the industry’s stance. For a comparison with international regulations, see Section 8: European Regulations on PFAS Chemicals and TFA.
Conclusion:
The US leads in legacy PFAS control but lags on TFA. Including TFA under federal PFAS definitions will be essential for future regulatory alignment. This current exclusion of TFA limits its coverage under key federal statutes and reflects a regulatory focus on compounds with demonstrated bioaccumulation and toxicity. Industry groups support this narrower definition, citing structural and toxicological distinctions, but emerging scientific evidence and international regulatory trends suggest that TFA may warrant closer scrutiny. As the regulatory landscape continues to evolve, future policy decisions may revisit the scope of PFAS definitions and the inclusion of compounds like TFA, particularly as more data becomes available on their environmental behavior and potential health impacts.
8. EUROPEAN REGULATIONS ON PFAS CHEMICALS & TFA
Introduction:
PFAS contamination in Europe originates from the use of pesticides, HFO/HFC refrigerants, industrial discharges, and wastewater treatment. TFA has been detected in 94% of EU water samples and in agricultural and beverage products. The sources of Trifluoroacetic Acid (TFA) contamination include PFAS Pesticides, F-gases, Industrial activities, and Wastewater Treatment Plants (WWTPs). Agricultural runoff allows over 30 PFAS pesticides approved in the EU to degrade into TFA, contaminating soil, groundwater, and crops. For example, flufenacet, a common herbicide, was applied to 2.9 million hectares of UK farmland in 2022.
This has a secondary impact on the food chain as TFA accumulates in crops. Studies found the EU wines had TFA levels of 122 μg/L in 2021 2024, which had increased from undetectable levels in 1988. Conventional grain products averaged 3x higher TFA levels than organic options. Refrigerants and coolants, including HFC and HFO F-Gases, release TFA precursors into the atmosphere, which deposit into the water systems via rainfall. Emissions from HFO-1234yf releases alone are expected to reach 47,560 tons/year by 2050. Studies of German rainwater have found a fivefold increase over 20 years, reaching as high as 335 ng/L, highlighting the concern.
Industrial activities, including pharmaceutical production, fluoropolymer production, and firefighting foam production, release TFA precursors as a secondary source of TFA. Furthermore, even Wastewater Treatment Plants (WWTPs) introduce TFA into the water system as TFA precursors in sewage, which convert to TFA during treatment, but cannot be removed with current treatment methods.
EU Regulations:
A universal PFAS restriction under REACH (decision post-2025) may include TFA. Given its persistence and toxicity, EU regulators and ECHA are preparing classification updates.
The European Union (EU) has developed a comprehensive and evolving regulatory framework to address the risks posed by PFAS to human health and the environment. The key elements of the European PFAS Regulation are based on four specific regulations. First, the REACH Regulation (EC 1907/2006) requires registration, evaluation, and restriction of chemicals, including PFAS. Several PFAS are listed specifically as substances of very high concern (SVHC), and some of these are subject to specific restrictions or bans. The Persistent Organic Pollutants (POPs) Regulation (EC 2019/1021) implements bans and restrictions on certain PFAS, including PFOS, PFOA, and PFHxS, in line with the Stockholm Convention.
The Drinking Water Directive (EU 2020/2184), which will take effect in January 2026, will establish a combined limit for total PFAS in drinking water at 500 ng/L. This directive will come into force across the EU, although member states can opt out of enforcing this value. Finally, Food and Water Regulations have set maximum levels for PFAS in foodstuffs under EC Regulation 2023/915. Separately, the levels of PFAS in water bodies are regulated under the Water Framework Directive (2000/60/EC).
Currently, TFA is not explicitly regulated under EU Regulations, as it is not among the 20 PFAS specifically regulated under the EU Drinking Water Directive. There are no dedicated limits in EU surface, groundwater, or drinking water either. However, the European Reference Laboratory has set a reporting limit for TFA in fruits and vegetables. The Dutch RIVM has made country-specific recommendations for TFA levels in drinking water of 2,200 ng/L; however, this is not legally binding at present. The regulations and their application to PFAS in general and TFA specifically are shown in the following chart:
With widespread contamination, ongoing sources of TFA contamination, and concerns about toxicity, there is a strong call for action within the EU and its regulatory bodies. Recent studies, which show TFA in 94% of EU drinking water samples with TFA concentration levels generally higher than other PFAS, highlight these concerns as an urgent issue. Meanwhile, PFAS pesticides and F-gases remain a major contributor to continued TFA contamination, inspiring regulators to target bans and restrictions to reduce the ongoing TFA contamination.
The outlook for EU regulation indicates that the EU is moving toward a universal restriction on PFAS, with a decision to be made after 2025. If successful, this would likely include phased bans and some exceptions for essential uses. While TFA is not specifically regulated, it is under increasing scrutiny due to its environmental persistence, widespread presence, and emerging evidence of toxicity.
Regulatory action is currently under active consideration, but no enforceable limits have been established yet. Despite concerns about TFA propagation, the inclusion of TFA in the 2026 total PFAS drinking water limit remains uncertain, and its regulation is expected to evolve in the coming years as scientific and political consensus builds.
Conclusion:
The EU leads in PFAS management but must close the TFA gap. Future regulation will likely extend total PFAS limits to include TFA, harmonizing EU and OECD definitions. Trifluoroacetic Acid (TFA), despite its environmental persistence and widespread detection, remains largely unregulated at the EU level. While some national guidelines and reporting thresholds exist, there are currently no binding limits for TFA in drinking water or the environment. Ongoing scientific assessments, including proposals to classify TFA as toxic to reproduction and environmentally persistent, suggest that regulatory attention is intensifying. Given the increasing evidence of contamination and potential health risks, future EU regulations are expected to expand in scope and stringency, potentially including TFA under broader PFAS restrictions in the years to come.
9. REMOVAL AND REMEDIATION METHODS FOR TFA REMOVAL FROM WATER
Introduction:
HFO refrigerants create TFA, which resists degradation. Current treatment methods are limited and expensive, with some
even generating TFA as a byproduct.
Regrettably, many of the mitigation methods for PFAS today actually generate TFA, another PFAS chemical, as a byproduct. Compounding the impact on TFA concentration levels in our water supply, there is currently no cost-effective water treatment method available to remove TFA at scale from drinking water. Thus, this “forever chemical” with a decades-long half-life, no filtration means, and numerous release sources from F-gases, pesticides, and industrial processes will continue to accumulate and build upon concentration levels in our drinking water.
Removal and Remediation Methods:
Traditional biological treatment and ozonation methods are ineffective in degrading TFA. Furthermore, high chloride and sulfate concentrations in drinking water compete with TFA for adsorption sites, reducing the efficiency of adsorption methods for TFA removal. Costs remain the most prohibitive challenge for the large-scale implementation of cutting-edge methods, such as electro-assisted systems. Several other new methods show promise; however, all are cost-prohibitive at scale. Ultimately, considering hybrid approaches that utilize more than one of these approaches may yield efficacy and meet cost objectives.
Industrial and Municipal Water Treatment methods under consideration include Ion Exchange Resins, Reverse Osmosis, Electro-Assisted Desorption, and Advanced Desorption. Specially designed resins, such as surface-defunctionalized activated carbon felt, adsorb TFA via electrostatic attraction and hydrophobic interactions. Cost and efficiency of this method are the primary challenges, although newer resins have demonstrated improved efficiency. Reverse osmosis systems can effectively block TFA molecules, with studies confirming complete retention in full-scale water treatment facilities.
However, operational costs and energy use limit their adoption. With Electro-Assisted Desorption, modified activated carbon is combined with low-voltage electrochemical cells to achieve greater than 85% TFA recovery using a sodium sulfate electrolyte. This trap-and-release approach will require adsorbent regeneration over multiple cycles.
The primary challenges and limitations of TFA removal and remediation methods are the exorbitant costs for treatment, selectivity issues where divalent ions, such as calcium or magnesium, interfere with TFA removal in RO and ion exchange systems, and the environmental persistence of TFA. With no known biodegradation pathway existing, a permanent removal or destruction method must be developed. The emerging solution is a two-pronged approach. First, utilizing a hybrid system combining RO with pre-treatment ion exchange to improve the cost-efficiency of the TFA removal. Policy driven source control targeting TFA precursors, such as PFAS pesticides and F-gases, could reduce environmental loading by 40-60%. But the critical gaps in this plan are also two-pronged. Technological challenges to developing a commercially viable water treatment solution, combined with a lack of political will to minimize or eliminate the use of TFA precursors, will exacerbate the accumulation of TFA.
Lyophilization (freeze-drying) is widely used in laboratory settings to remove TFA from aqueous solutions; however, its practicality for large scale industrial water treatment is limited by several significant factors. First, lyophilization achieves incomplete removal of bound TFA. Free TFA ions can be removed, but TFA that is directly associated with other compounds often remains even after freeze-drying. The scale-up challenges are significant because lyophilization is inherently slow and is generally deployed in low-throughput operations. Furthermore, industrial-scale equipment would be very expensive and require a significant amount of energy input. Additionally, TFA is corrosive, especially at higher concentrations, which can lead to damage to the lyophilization components.
Finally, this process introduces significant operational complexity, including mixed solvent issues such as solvent bumping, cross contamination, and the need for deep vacuum and low temperatures, which would further complicate the scaling-up process. The most promising solution is combining reverse osmosis with ion exchange resins. The effectiveness and limitations of these resins become crucial factors. The technical and operational challenges associated with ion exchange resins, particularly at scale, are substantial. First, these resins have limited effectiveness for TFA because it is a small, highly water-soluble molecule, making it more difficult for conventional resins to capture compared to longer-chain PFAS.
Additionally, the presence of other ions, such as chloride, sulfate, and nitrate, competes with TFA for binding sites on the resin, significantly reducing its efficiency. Over time, the resins become fouled by organic matter, such as iron or manganese, which can clog or diminish the resin’s efficiency.
Furthermore, TFA’s acidic nature also contributes to resin degradation. This resin degradation will require significant maintenance activities and costs. As the resin accumulates captured TFA, it must be regenerated, which yields a secondary waste stream of a brine containing concentrated TFA. This waste stream would also require further treatment and safe disposal. Incineration is the primary means of treating this waste stream, although it is costly and may release PFAS into the environment. In addition to high first- and maintenance costs, ion exchange resins also have high operational costs. Although the hybrid solution reduces the first cost by 20-40%, operational costs remain high. Finally, the resin solution is tailor-made, and not all resins are equally effective for different types of PFAS. The solution must be customized to address the specific contamination in the water and may need to be adjusted as water contamination evolves.
TFA is particularly challenging for ion exchange resins due to its strong binding affinity. TFA is highly acidic due to the strong electronegativity of the fluorine atoms, making it resistant to displacement during standard regeneration cycles. Consequently, weaker acids only partially displace TFA from resins during the regeneration process, requiring stronger bases for complete removal. Typical regenerating agents, such as NaCl brine, are limited to 50% TFA elution efficiency and require specialized chemicals, such as concentrated NaOH or methanol mixtures, for effective regeneration, increasing costs and complexity.
The economic and operational barriers to overcome include 2-3x higher chemical consumption, a large amount of waste volume, which is difficult to manage with TFA-laden brine of only 1-5%, and very high energy costs to achieve thermal regeneration at or above 300°C. As discussed above, resin degradation resulting from repeated exposure to harsh regeneration conditions requires significant maintenance and resin replacement costs.
The final consideration for ion exchange resins is the disposal method. As discussed previously, incineration is a common method. Still, it requires significant energy to achieve temperatures of 800-1200°C and can release toxic byproducts, including Hydrogen fluoride, TFA from partial reformation, and Perfluorocarboxylic acids (PFCAs). Landfilling is another approach; however, TFA’s long-term persistence means that it can persist in landfills for centuries. Due to its water solubility, TFA can also leach into landfill leachate, posing a risk of groundwater contamination if it’s not fully contained.
Underground injection into Class 1 Wells is another approach to isolate TFA waste. Still, it raises risks of aquifer contamination, capacity limitations of Class 1 wells, and potential geological instability that may provide pathways for TFA contamination of surface waters.
All removal and remediation methods today face significant challenges in terms of cost, scalability, and recontamination. The accumulation effect is making the issue more urgent all the time, so quick action, investment in large-scale water treatment solutions, and reducing the TFA load at the source are all required.
Conclusion
High energy costs, selectivity challenges, and the disposal of TFA waste prevent large-scale deployment; however, a dual strategy combining technological innovation and source control—offers the best approach to eliminating TFA. Hybrid RO + IXR systems, combined with upstream emission bans, could reduce TFA loads by 40–60%. Nevertheless, no existing method fully eliminates TFA at scale. Source prevention remains the most viable long-term strategy for curbing accumulation and protecting global water resources. While advanced technologies, such as reverse osmosis, ion exchange resins, and electro-assisted desorption, show promise, each faces limitations in terms of scalability, cost, and operational complexity. Hybrid systems combining these methods may offer improved efficiency, but they still require substantial investment and infrastructure. Given the lack of effective large-scale solutions and the ongoing accumulation of TFA in water systems, future efforts will likely need to focus on both technological innovation and upstream source control to mitigate long-term impacts.
APPENDIX 1 – PFAS & TFA: REGULATORY LANDSCAPE AND STRATEGIC REFRIGERANT CHOICES
Regulatory Definitions of PFAS
Health and Environmental Risks of TFA
• Acute human toxicity: Low to moderate
• Chronic ecological impact: High
• Persistent, mobile, and bioaccumulative
• Accumulates in water systems and soil
• Difficult to remediate
• Considered a ‘planetary boundary threat’ due to long-term environmental persistence
Strategic Advantages of Natural Refrigerants
• No PFAS-related liabilities
• Zero ozone depletion potential (ODP)
• Low global warming potential (GWP)
• Aligns with sustainability and ESG goals
• Reduces regulatory and reputational risk
• Examples: CO₂ (R-744), Ammonia (R-717), Propane (R-290)